LiliF® Listeria Real-time PCR Kit
Listeria is a genus of bacteria that, until 1992, contained 10 known species, each containing two subspecies. As of 2014, another five species were identified. Named after the British pioneer of sterile surgery Joseph Lister, the genus received its current name in 1940. Listeria species are gram-positive, rod-shaped, and facultatively anaerobic, and do not produce endospores. The major human pathogen in the genus Listeria is L. monocytogenes. It is usually the causative agent of the relatively rare bacterial disease listeriosis, an infection caused by eating food contaminated with the bacteria. Listeriosis can cause serious illness in pregnant women, newborns, adults with weakened immune systems and the elderly, and may cause gastroenteritis in others who have been severely infected.
This LiliF™ Listeria Real-time PCR Kit is designed as a probe-based real-time PCR product targeting the specific sequence in the hly gene of Listeria monocytogenes. It uses the highly efficient Hot Start PCR Enzyme to maximize the detection performance of the product. This product is suitable for testing the mixed Listeria monocytogenes bacteria in the specimens with high sensitivity and specificity.
• LiliF™ Listeria Real-time PCR Kit is a 5 'Nuclease Assay Real-time PCR product.
• This product specifically tests Listeria monocytogenes for DNA from food-borne samples and microorganism culture. The validity of the test can be verified by providing Control DNA for the verification of the reaction.
• The target gene is the hly gene of Listeria monocytogenes. It can specifically detect only Listeria monocytogenes among other Listeria.
• This product is a kit for the detection of harmful microorganisms in various foods and livestock products.
•It is a product for qualitative analysis by real-time PCR test for the presence or absence of Listeria monocytogenes in microorganism culture and microbial culture.
| No. | Contents | 50 Tests/Kit |
|---|---|---|
| 1 | 2X qPCR Master Mix Solution | 520 μl x 1 vial |
| 2 | Listeria Detection Premix | 260 μl x 1 vial |
| 3 | Positive Control | 25 μl x 3 tubes |
| 4 | DNase/RNase Free Water | 1 ml x 1 tube |
Performance
Sensitivity
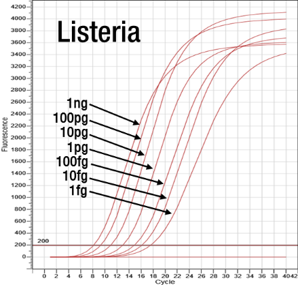
Seven steps of dilution of the plasmid DNA including the target gene (hly gene) in the Listeria monocytogenes was confirmed to be positive using the PCR examination.
Interpreting Result
Interpretating Samples
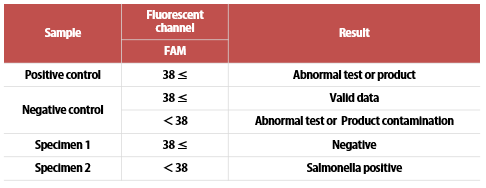
LiliF™ Listeria Real-time PCR Kit is designed as a Probe Real-time PCR product with a specific sequence in the hly gene of Listeria monocytogenes and can interpret the results through quantitative analysis of the fluorescent channel.
| Product | Cat.No | Capacity | inquire | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| G-spin™ Total DNA Extraction Mini Kit Best |
|
|||||||||