LiliF® COVID-19 Real-time RT-PCR Kit
There are four genes in the Coronavirus family. Those are known to alpha, beta, gamma, and delta. Alpha and beta corona viruses can cause illness in both humans and animals, whereas others, such as gamma and delta coronaviruses, only infect animals.
The new coronavirus (COVID-19) belongs to beta and is one of the new infectious corona viruses that infects the human body as a pathogen of mass pneumonia that occurred in Wuhan, Hubei, China in December 2019. It is very important to diagnose an infection quickly, because there are no vaccines or antivirals approved for prophylactic or therapeutic purposes.
Accordingly, by detecting the RdRp and E genes that are mainly used in Korea as of February 2020, and the N genes, which are recently used as a standard for testing in the US CDC, the diagnostic reliability is increased, and only template RNA is added so that anyone can use them. Mastermix type of product can increase the speed, accuracy and convenience of molecular diagnosis of new corona viruses.
• LiliF™ COVID-19 Real-time RT-PCR Kit can detect the new coronavirus using probe method of Real-time RT-PCR, through the reacting of the specific primer and Fluorescent probe in sample.
• This product is provided with a quantitative aliquot of a reagent, primer and probe that performs Real-time RT-PCR easily to detect new coronaviruses. The user can immediately experiment by adding RNA extracted from the sample.
• LiliF ™ COVID-19 Real-time RT-PCR Kit can detect RdRp and E gene, markers for detecting new coronaviruses. Also, N gene suggested by the US CDC and RNaseP gene which can confirm the validity of all test reactions are adopted and designed for simultaneous detection.

• Sputum in the lower respiratory tract, Bronchoalveolar lavage fluid (BAL), or nasopharyngeal swab (NS) and oropharyngeal smear (Oropharyngeal swab, OS) simultaneously collected from the upper respiratory tract. In vitro diagnostic medical devices that help diagnose new coronavirus infection (COVID-19) by qualitatively detecting RdRP genes, E genes and N genes of the new coronavirus (2019-nCoV) from the sample
| No. | Contents | 50 tests/kit |
|---|---|---|
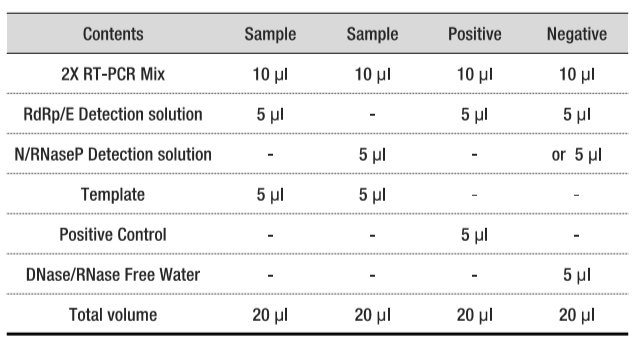
| 1 | 2X RT-PCR Mix | 1120 μl x 1 tube |
| 2 | RdRp/E Detection Solution | 280 μl x 1 tube |
| 3 | N/RNaseP Detection Solution | 280 μl x 1 tube |
| 4 | Positive Control | 450 μl, 1 tube |
| 5 | DNase/RNase Free Water(Negative Control) | 1 ml x 1 tube |
| Product | Cat.No | Capacity | inquire | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Miracle-AutoXT Automated Nucleic Acid Extraction System Best |
|
|||||||||
| Patho Gene-spin™ DNA/RNA Extraction Kit Best |
|
|||||||||