Human RDT
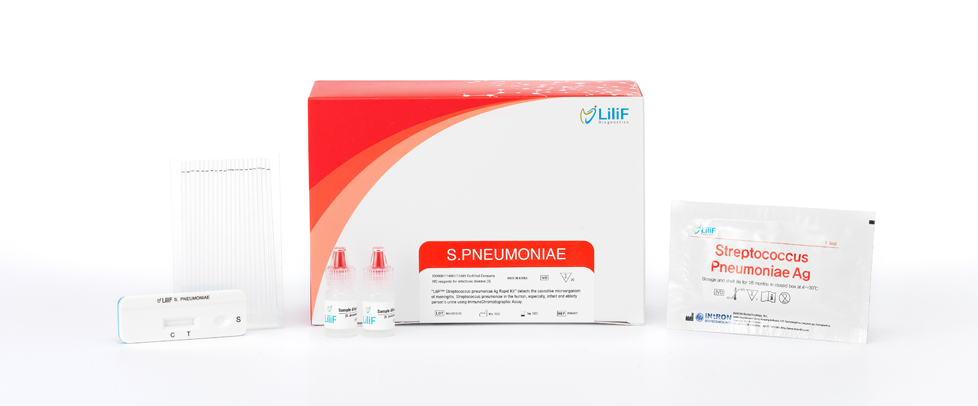
LiliF® Streptococcus pneumoniae Ag Rapid kit
| Cat.No |
Capacity |
Inquire |
| IRH41631 |
1 test X 20 / kit |
 |
PRODUCT INFORMATION
Background Information
Pneumonia is not a big problem when it occurs in healthy people, but it is a very dangerous disease in children and the elderly. Therefore, it is important to diagnose the exact causative agent of pneumonia using an appropriate test method and to start effective antibiotic treatment at an early stage. The most common bacterial pneumonia causative agent in Korea is Streptococcus pneumoniae, accounting for approximately 30-40% of the total. Diagnosis of infection caused by Streptococcus pneumoniae is by traditional culture method using sputum or blood, but it must be diagnosed before antibiotic administration, and the reliability is low. However, immunochromatography using urine during antigen testing for Streptococcus pneumoniae has high sensitivity because it can detect C-polysaccharide in the cell wall for all species of Streptococcus pneumoniae.
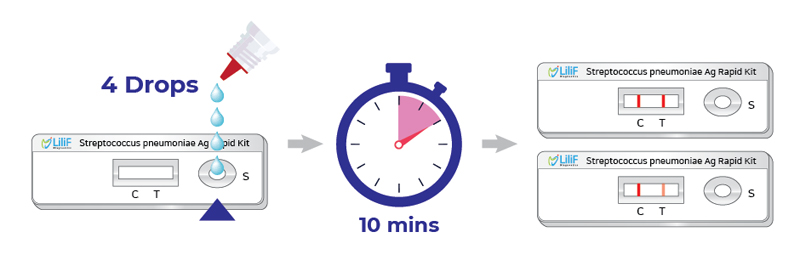
LiliF™ Streptococcus pneumoniae Ag rapid kit uses ImmunoChromatographic Assay to diagnose pneumococcus, the causative agent of pneumonia, which is a major disease. can be read within 10 minutes).
Intended Use
• In vitro diagnostic medical device manufacturing license In vitro No. 22-80 - High-risk infectious agent gene test reagent
• An in vitro diagnostic medical device that helps confirm pneumococcal infection by qualifying the Streptococcus pneumoniae antigen (C-polysaccharide) in the urine sample of a patient with suspected pneumonia using ImmunoChromatographic Assay (ICA)
Kit Contents
| No. | Contents | 20 tests/kit |
|---|
| 1 | Test device | 20ea |
| 2 | Sample diluent | 2ea |
| 3 | Dropperd | 20ea |
| 4 | IFU | 1ea |
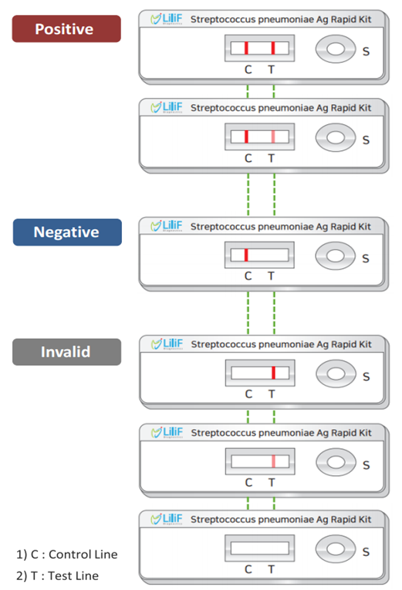
Interpretation
Related Product
관련제품 Product,No,Capacity,Inquire list
| Product |
Cat.No |
Capacity |
inquire |
List